Project Management
Tasks, Kanban, Sprint, Gantt, Calendar — inline editing, story points, dependencies. The thing Jira makes you wait three releases for.
See project mgmt →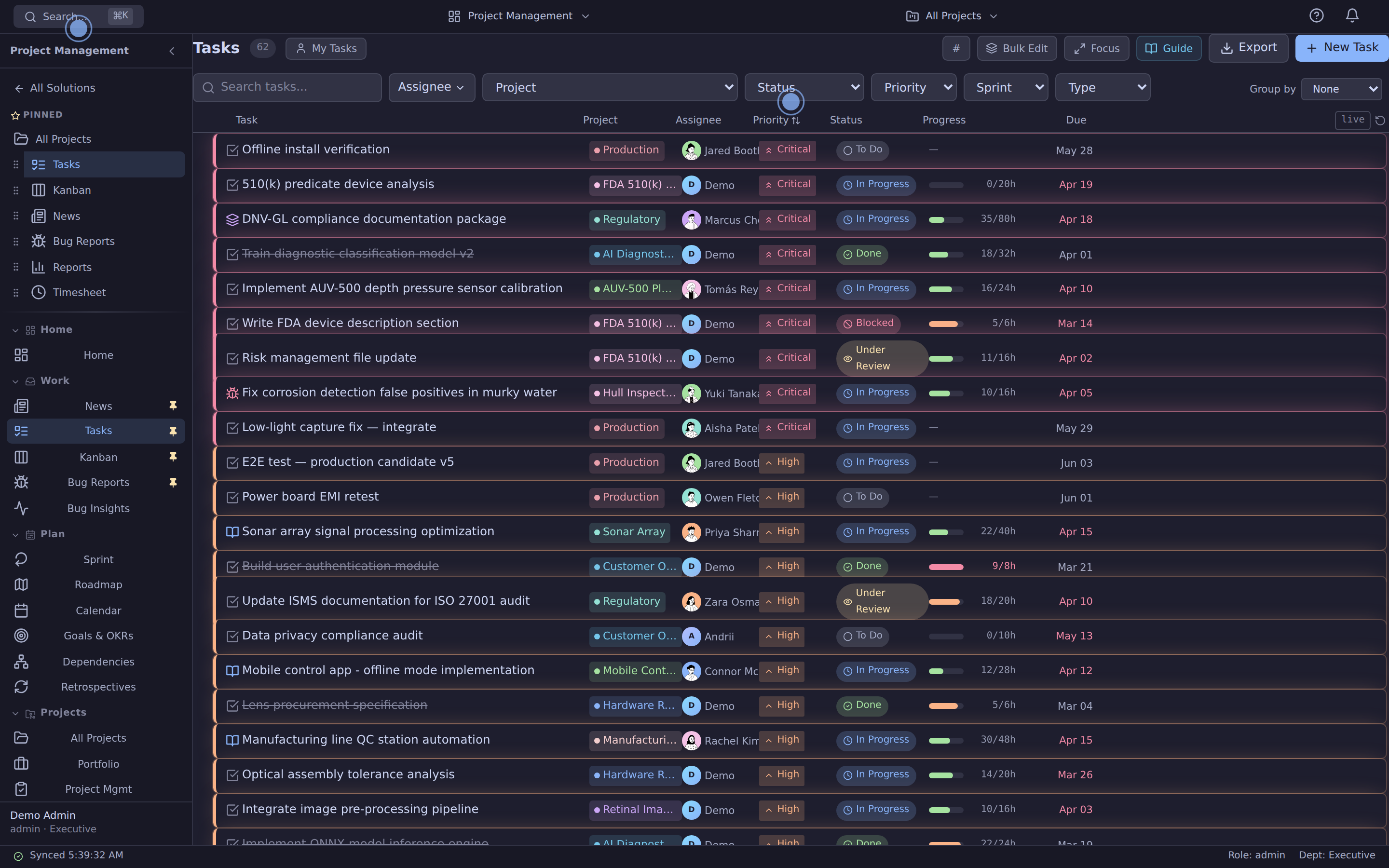
Project management, QMS, ERP, HR, and documentation — one platform, one login, one bill. For engineering teams tired of stitching together Jira, Notion, BambooHR, QuickBooks, and four others that don't talk to each other.
Free tier up to 5 users · 14-day Pro trial · Self-hostable on your VPS

50+
Integrated panels
8 in 1
Tools replaced per team
1 bill
Every module, no per-module fees
100%
Tenant-isolated by row
Real screenshots from a live demo tenant. Cycles every five seconds — click a tab to jump, hover to pause.
Tasks, Kanban, Sprint, Gantt, Calendar — inline editing, story points, dependencies. The thing Jira makes you wait three releases for.
See project mgmt →
Scroll this into view and watch the stack collapse — every tool replaced by one login, one schema, one invoice. Row-level tenant isolation, not workspaces.
platform · login · bill · database
Stop paying for Jira + Notion + Greenlight Guru + BambooHR + QuickBooks + Slack + Confluence + Asana. Engineering teams ship faster when their tools talk to each other.
Tasks, Kanban, Sprint, Roadmap, Gantt, Calendar, Bugs, Goals, Portfolio. Inline editing, drag-drop, dependencies, story points, swimlanes — all the things Jira makes you wait for.
Replaces: Jira · Asana · Linear
CAPA, NCR, Change Control, Audits, Complaints, Risk Register, Design Reviews, Traceability, Test Mgmt, Training. ISO 13485 audit-ready with signed change control.
Replaces: Greenlight Guru · MasterControl
Purchasing, Vendor Tracker, Budget, Timesheet, Support — POs to projects, time to invoices, vendors to the risk register.
Replaces: QuickBooks (light) · Procurify
Employees, Leave, Onboarding, Compensation, Benefits. Org chart with manager hierarchy — every panel knows who you are.
Replaces: BambooHR · Personio
Documents, Guidelines, Wiki, Task Dump, Automation, Releases. Versioned, searchable, and linked to the work they describe.
Replaces: Confluence · Notion · SharePoint
Units, Unit Genealogy, Lab & Equipment. Track every serialized unit from BOM to deployment — built for companies that ship physical hardware.
Replaces: Custom-built spreadsheets
Pick your team size and the tools you currently pay for. We'll show what you'd save by consolidating onto Azora Pro at $39/user/month.
Per-seat prices below are the public list price for each tool's mainstream team tier.
Math assumes your team buys each selected tool's listed seat tier. Real-world stacks usually include vendor-specific discounts and consultant fees we haven't modelled. Azora Pro is uncapped users and modules during open beta.
The screenshots are already dark — here they sit in their natural habitat, lit by ambient glow. The sprint burndown is rebuilt from persisted daily snapshots, not faked from current state at render time.

Every record carries an account_id. No folder gymnastics, no shared workspaces. Your data is yours, full stop.
TOTP secrets are AES-256-GCM at rest with HKDF-derived keys. Other secrets (license keys, integration tokens) move to the same envelope in Q3 2026.
Signed change control, per-tenant audit log, electronic signatures designed around 21 CFR Part 11. The paperwork your auditor expects, generated automatically.
Scheduled rules with trigger → condition → action. Notify on overdue, auto-assign by department, escalate blocked tasks — built into the database, not bolted on.
Run it on your own VPS for compliance reasons, or let us host. Same code, same database schema, your call.
Postgres under the hood. Standard schema. Self-service CSV export ships Q3 2026; manual export available today on request.
Per-user jwt_issued_at column. Logout, suspension, or password change bumps it; every live token for that user invalidates instantly. Most SaaS doesn't bother.
RAID log, Decision Rights, Manager Roll-up, 1:1 prep, Skip-level visibility, Weekly Status. The framework most teams pay $99/seat for separately. Read more →
Email us with your team name and seat count. We'll provision a seeded tenant with sample data and send you a login within 24 hours. Every module unlocked, no demo gating.